Columbia University
Irving Medical Center
Neurological Institute
710 West 168th Street, 3rd floor
(212) 305-1818
TaubCONNECT Research Perspectives:
August 2019
2: » Live Imaging of ESCRT Proteins in Microfluidically Isolated Hippocampal Axons
Neuroinflammation in Frontotemporal Lobar Degeneration Revealed by 11C‐PBR28 PET

William C. Kreisl, MD
Animal studies have demonstrated that deletion of genes associated with inherited forms of frontotemporal lobar degeneration (FTLD) cause altered neuroimmune responses, suggesting that neuroinflammation may play a pathogenic role in FTLD. Therefore, positron emission tomography (PET) scans using radioligands specific to active components of the neuroinflammatory process could be useful for monitoring disease‐specific brain changes in living patients with FTLD.
Using the second‐generation 18‐kDa translocator protein (TSPO) radioligand 11C‐PBR28, previous studies by Dr. William (Chuck) Kreisl and colleagues found strong correlations between brain uptake and the clinical severity or progression of Alzheimer's disease (AD). Another study found that the distribution of 11C‐PBR28 binding colocalized to neurodegeneration in different clinical subtypes of AD. Building on this work, the present study by Kreisl and colleagues used 11C‐PBR28 PET imaging to determine whether levels of the inflammation specific biomarker TSPO are increased in FTLD patients, within their relevant affected brain areas.
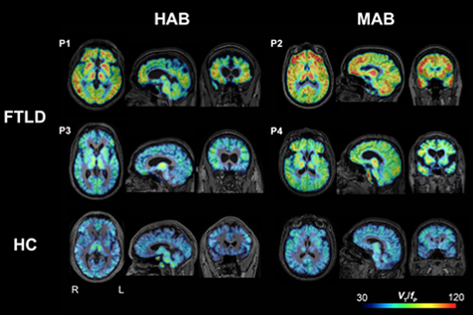
Figure 1: Parametric images of 11C‐PBR28 total distribution volume corrected by plasma‐free fraction (VT/fP) in four patients with frontotemporal lobar degeneration (FTLD) (P1, P2, P3, and P4) and two representative healthy control subjects (bottom row). When visually compared to healthy control subjects, FTLD patients had greater 11C‐PBR28 binding in frontotemporal cortices and other brain areas. Abbreviations: HAB, high‐affinity binder; MAB, mixed‐affinity binder; HC, healthy control.
As recently published in the Annals of Clinical and Translational Neurology, Kreisl and colleagues conducted 11C‐PBR28, 18F‐FDG, and 11C‐PIB brain PET scans, as well as magnetic resonance imaging (MRI), in four FTLD patients and 22 healthy controls. 11C‐PBR28 scans revealed that all FTLD patients showed increased TSPO binding versus controls. Regions of increased 11C‐PBR28 binding in FTLD patients corresponded with the expected neurodegeneration predicted by clinical phenotype, as well as MRI and 18F‐fluorodeoxyglucose (FDG) PET scans. Amyloid burden, as measured by 11C‐PIB, was not increased in any gray matter region in FTLD patients, consistent with the non-amyloid pathology of FTLD. The results also confirm that the simplified ratio method of 11C‐PBR28 PET developed for use in AD can be extended for use in FTLD.
William C. Kreisl, MD
Boris and Rose Katz Assistant Professor of Neurology (in the Taub Institute)
wck2107@cumc.columbia.edu
Live Imaging of ESCRT Proteins in Microfluidically Isolated Hippocampal Axons
 |
 |
 |
||
| Clarissa Waites, PhD | Ulrich Hengst, PhD | Veronica Birdsall |
In neurons, efficient protein clearance is crucial not only for maintaining functional synapses and therefore effective neural networks, but also for warding off neurodegeneration due to the accumulation of misfolded proteins. The lysosome is the cellular organelle responsible for breaking down biomolecules, including protein aggregates, mitochondria, and vesicles. One of the major conduits for trafficking proteins to the lysosome is the endocytic pathway, which is vital for protein degradation in organisms from yeast to humans [1-4]. This pathway mediates the degradation of membrane-associated proteins via Rab GTPases and the endosomal sorting complex required for transport (ESCRT) machinery [5, 6]. This series of protein complexes recruits ubiquitinated cargo for degradation, and forms multivesicular bodies (MVBs) for the delivery of cargo to lysosomes.
It has recently emerged that the ESCRT pathway is involved in maintaining multiple aspects of nervous system function, from neuronal morphogenesis to synaptic plasticity and vesicle secretion [7,8]. These findings have led to heightened interest in the study of this pathway in neurons. Such interest has only intensified as ESCRT pathway dysfunction is increasingly linked to neurodegenerative disease etiology. For instance, mutations in the ESCRT-III protein CHMP2B cause frontotemporal dementia and amyotrophic lateral sclerosis, and deletion of other ESCRT components (i.e. HRS, TSG101) in mouse models induces profound neurodegeneration [9-14]. However, it has traditionally been difficult to study the ESCRT pathway in axons and presynaptic terminals due to the low endogenous expression of ESCRT proteins in these compartments. In this study, Drs. Clarissa Waites and Ulrich Hengst, along with first author Veronica Birdsall, a PhD candidate in the Neurobiology and Behavior Program, develop a methodology to visualize the localization and dynamic behavior of ESCRT proteins in axons of cultured neurons. They achieve this through lentiviral transduction of fluorescently tagged ESCRT proteins, coupled with microfluidic axon isolation and live cell imaging in rat hippocampal neurons. Birdsall et al. provide a detailed protocol for this new strategy in a chapter of Methods in Molecular Biology.
Clarissa Waites, PhD
Associate Research Scientist in the Department of Pathology and Cell Biology
cw2622@cumc.columbia.edu
Ulrich Hengst, PhD
Associate Professor of Pathology and Cell Biology (in the Taub Institute for Research on Alzheimer's Disease and the Aging Brain
uh2112@cumc.columbia.edu

