Columbia University
Irving Medical Center
Neurological Institute
710 West 168th Street, 3rd floor
(212) 305-1818
TaubCONNECT Research Perspectives:
December 2015
» #2 Telomere Longitudinal Shortening as a Biomarker for Dementia Status of Adults With Down Syndrome
 |  |
| Giuseppe Tosto, MD, PhD | Richard Mayeux, MD, MSc |
Alzheimer's disease (AD), the leading cause of dementia in the elderly, is characterized by progressive cognitive impairment, predominantly in the memory domain, at least in the early stages. Most studies assess the disease as a single phenotypic outcome, neglecting its heterogeneity. In fact, motor and psychiatric manifestations, in particular extrapyramidal signs (EPS) and hallucinations, are frequently observed. This is an important aspect of the disease, as patients with EPS tend to have a faster rate of cognitive decline compared to those without EPS. These clinical signs are present in AD, as well as in dementia with Lewy bodies (DLB) and in Parkinson's disease (PD), and these three entities also share neuropathological features. While AD, DLB, and PD are currently defined as individual disease entities, it remains possible that they actually reflect a continuum of a single spectrum disorder. Previous work by Taub faculty members Drs. Giuseppe Tosto and Richard Mayeux demonstrated that the pattern of EPS (i.e. rigidity, tremor, limb bradykinesia, etc.) in AD resembles what is commonly observed in PD.
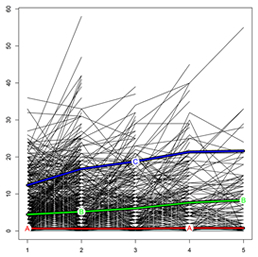
Fig. 3. KmL plot. Each line defines a single patient's UPDRS score progression during follow-up. The "A" line represents the cluster with no/low EPS load, the "B" line the cluster with medium EPS load, and the "C" line the cluster with high EPS load. X-axis, years of follow-up; Y-axis, UPDRS total score.
More recently, in a study published in the Journal of Alzheimer's Disease, Drs. Tosto, Mayeux, and colleagues from Washington University examined the progression of EPS in 3,502 AD patients from the National Alzheimer's Coordinating Center (NACC) dataset, capitalizing on the rich, longitudinal phenotyping of this cohort. Progression of extrapyramidal signs was determined by drawing sub-clusters of escalating severity, employing a novel implementation of k-means algorithm specifically designed for longitudinal data. Three clusters of EPS progression were identified: none/low, medium, and high EPS burden. Individuals belonging to the high EPS cluster had greater cognitive and neuropsychiatric impairment (particularly hallucinations) as compared to those with none/low and medium EPS. In addition, despite similar AD-pathology across the three clusters, the high EPS cluster had higher likelihood of being diagnosed with DLB.
This study provides further evidence that the neuropathological underpinning of AD differs for distinct phenotype patterns. It also demonstrates that k-means clustering analysis is a useful tool for characterizing the heterogeneous nature of AD and other conditions, especially when dealing with non-normal data as EPS usually present.
Giuseppe Tosto, MD, PhD
Postdoctoral Research Scientist (in the Taub Institute and the Gertrude H. Sergievsky Center)
gt2260@cumc.columbia.edu
Richard Mayeux, MD, MSc
Gertrude H. Sergievsky Professor of Neurology, Psychiatry and Epidemiology (in the Gertrude H. Sergievsky Center and in the Taub Institute)
rpm2@cumc.columbia.edu
Telomere Longitudinal Shortening as a Biomarker for Dementia Status of Adults With Down Syndrome
 |  | |
| Nicole Schupf, PhD, MPH, DrPH | Edmund C. Jenkins, PhD |
Telomeres are stretches of highly conserved TTAGGG repeats on the ends of chromosomes. These telomere sequences are not fully replicated during the normal process of cell division and thus become shorter with each subsequent cell cycle, eventually leading to the inability to replicate. Reduced telomere length has been associated with cellular aging, old-age associated neurodegenerative diseases, apoptosis, tumorigenesis, heart disease, and stress, as well as a host of psychosocial, behavioral, and environmental factors. Older individuals have shorter telomere lengths, but there is wide variation among similarly aged individuals. There has been considerable interest in telomere length as a biomarker associated with the development of Alzheimer's disease (AD) and while the preponderance of evidence indicates that short telomere length is a risk factor associated with the development of mild cognitive impairment (MCI) or AD, results have been mixed regarding the strength of this association.
Taub faculty member Dr. Nicole Schupf joined work led by Dr. Edmund Jenkins and colleagues at the New York State Institute for Basic Research in Developmental Disabilities and the Kennedy-Krieger Institute/Johns Hopkins University studying the association between telomere length and clinical stage of AD in adults with Down syndrome (DS), published this month in the American Journal of Medical Genetics: Neuropsychiatric Genetics. Adults with Down syndrome have a dramatically increased risk for AD, with virtually all adults developing its key neuropathological features by 40 years of age. The high levels of AD neuropathology in adults with DS have been attributed, at least in part, to triplication and overexpression of the gene for amyloid precursor protein (APP) located on chromosome 21. By age 70, estimates indicate that 90 percent of individuals with Down syndrome will have developed AD, although there is wide variation in age at onset of dementia, ranging from under 40 to over 70 years of age. Recognition of early AD-related changes is especially difficult for adults with DS because of their variable levels of pre-existing cognitive impairments. Thus, development of informative biomarkers for early diagnosis of dementia in DS are critical for differential diagnosis and effective intervention when concerns first emerge.
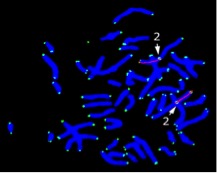
Figure 1. Metaphase
In previous cross-sectional studies, our group found a very strong relationship between shorter telomere length in T lymphocytes and the presence/absence of cognitive changes similar to mild cognitive impairment (MCI) in the general elderly population. The present study supported those findings by showing that, over time within the same individuals, telomeres become shorter with onset of MCI or dementia in adults with DS (MCI-DS). We used PNA (peptide nucleic acid) probes for telomeres on Chromosome 1 and Chromosome 2p and included the chromosome 2 centromere as an “internal standard” expected to be unaffected by aging or dementia status. Telomere length was examined in samples from the same individuals collected prior to and following development of MCI-DS or dementia (Figure 1: FISH preparation of a metaphase from short-term lymphocyte cultures showing telomeres). Mean interval between assessments was just under 3 years. Shortening of telomere length was observed in all individuals, with a mean decrease in telomere length of 42% for telomeres on Chromosome 1 and 43% for telomeres on Chromosome 2p. Comparisons with previous cross-sectional findings indicated that telomere lengths prior to clinical decline were similar to those of other adults with DS who did not show indications of MCI-DS while telomere lengths following cognitive decline were comparable to those of other adults with DS who had developed MCI-DS or dementia. These findings indicate that telomere length has significant promise as a biomarker of clinical progression of AD for adults with DS. However, samples have been quite small in all our studies and further longitudinal studies of larger samples are needed to validate these findings and determine if and how factors affecting AD risk also influence these measures of telomere length.
Nicole Schupf, PhD, MPH, DrPH
Taub Institute for Research on Alzheimer's disease and the Aging Brain
Ns24@cumc.columbia.edu
Edmund C. Jenkins, PhD
Institute for Basic Research in Developmental Disabilities
ed.jenkins@opwwdd.ny.gov

